Type 1 diabetes exists on a spectrum. Binder might represent the far end of one side, Coleman, McLeod, and Pokerznik the other. But the bottom line is that if anyone slips up, they can die, because even today’s most sophisticated medical devices still cannot match the precision of the tiny biological machines that come stock in the human pancreas: beta cells. That is why scientists have for decades tried to find a way to replace them.
(Eating too much of this favorite food could increase your diabetes risk)
An islet of Langerhans from the pancreas is seen in this colored scanning electron micrograph. This structure is composed of clumps of secretory cells that primarily release glucagon and insulin (blue), the hormones responsible for controlling blood sugar. Lennart Nilsson, Boehringer Ingelheim International GMBH, TT/Science Photo Library
A breakthrough decades in the making
The origins of the Vertex breakthrough trace back to one night in 1991, when Doug Melton, a 38-year-old professor of developmental biology at Harvard University, was studying the eggs of African clawed frogs in the basement of the Biological Laboratories. Melton was searching for the genes that tell the early stem cells of a single fertilized egg to transform into specialized cells. Then he got a call from his wife, Gail: Sam, their six-month-old son, was vomiting. Melton raced out of the lab. In the emergency room of Boston Children’s Hospital, he and Gail would learn their son had type 1 diabetes.
After the diagnosis, Melton gathered everyone in the lab—all 25 to 30 of his colleagues—and told them he was transitioning away from their research on frogs. Harvard had agreed to build Melton a new lab so that he could pursue a new mission: a cure for type 1 diabetes.
He wasn’t completely starting over. His work with the cells of frog embryos suggested a way forward. Those early stem cells are like biological blank slates: With the right stimuli, they can turn into any cell in the body. Melton just had to figure out the stimuli to coax human stem cells into functioning beta cells. He was already, as he puts it, “a painter, but now I was told what the subject of the painting had to be.”
Melton told me recently he had no idea how difficult the research would become, which he’s grateful for. If he’d known, he might’ve quit. “It seemed to me a simple idea,” he says: Engineer the cells his son had lost, then put them back in, all the while preventing the immune system from destroying the cells. He told his wife it would take him three to four years.
Between 1990 and 1999, Melton zipped past that three-to-four-year deadline and then zipped past it again. Melton wasn’t the only researcher looking into cell therapies, but they were each working in isolation. Back then, the scientific community was more preoccupied with trying to understand the mechanism by which insulin is secreted, not how to make the cells that do the secreting.
Then there was a breakthrough that cast their work into a new light. In a clinical trial between 1999 and 2000, University of Alberta doctor James Shapiro transplanted islet cells from cadavers into seven patients with severe type 1 diabetes. Other doctors had tried similar procedures with marginal success, but Shapiro developed a new protocol. He sourced healthy islets from multiple cadavers, processed the cells in a novel solution, and suppressed patient immune systems with steroid-free drugs. (Steroids tended to backfire in patients, making them insulin resistant and damaging their beta cells.)
Every patient quickly began producing enough of their own insulin that they no longer needed to inject it, and they continued producing insulin at follow-up more than a year later.
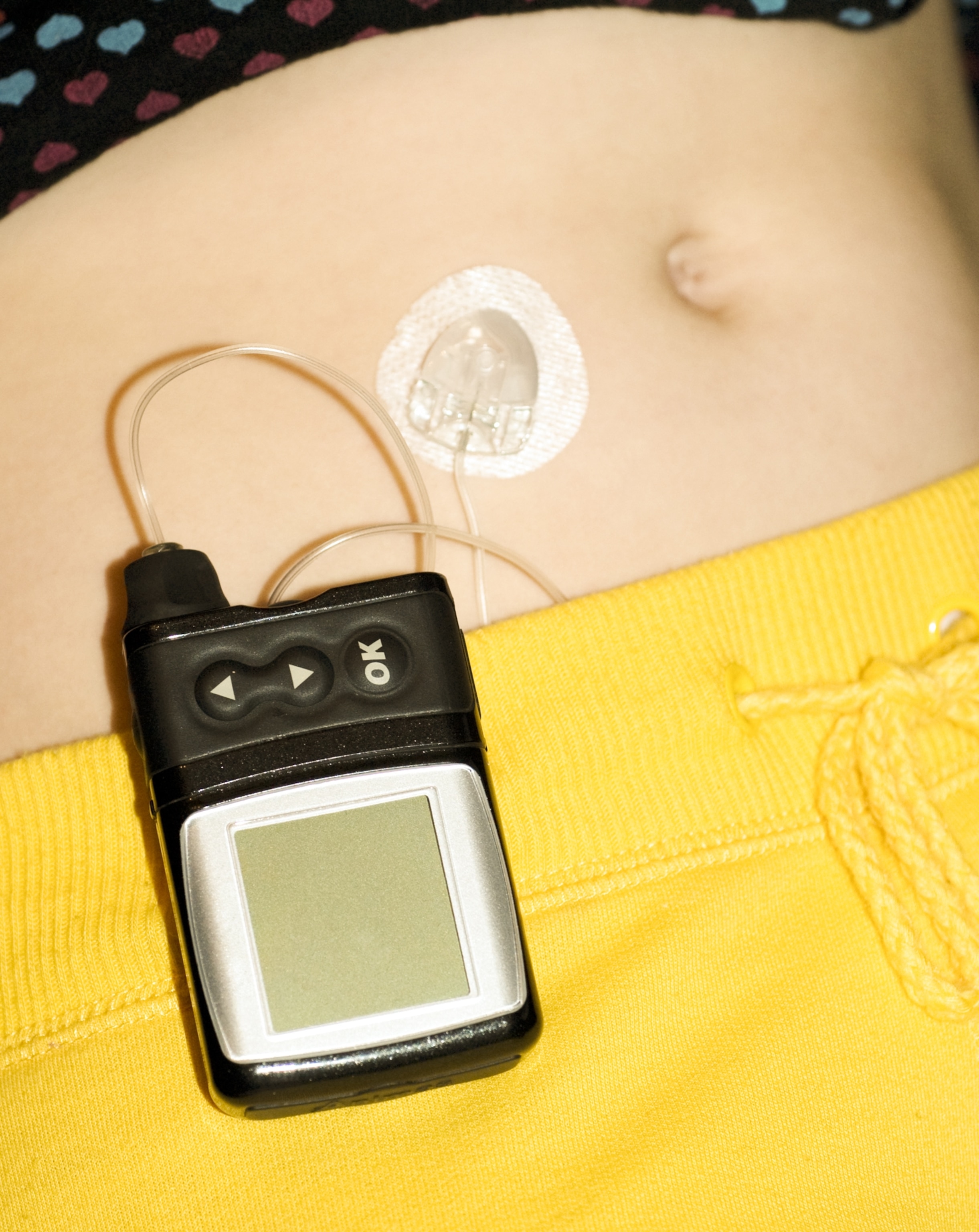
Insulin pumps help modern-day type 1 diabetics control their blood sugar. In the near future, stem cell therapies could replace the need for them.
Chu Montpellier, Phanie/Science Photo Library (Top) (Left) and Scott Camazine, Science Photo Library (Bottom) (Right)
Pokerznik was Shapiro’s second patient in the trial. “It was hard for me to even wrap my mind around,” she says, “because I’d been diabetic for so long.” Later, Shapiro also performed the procedure on McLeod and Coleman. The women say the therapy liberated them from the minute-by-minute calculations they had to make when managing their disease using pharmaceutical insulin. It also saved them from the constant crashes. “It changed a lot,” Coleman says. “Really, for me,” McLeod says, “there was no other option.”


