Type 1 diabetes is typically associated with more significant reductions in bone density than type 2 diabetes [4]primarily due to lower serum IGF-1 levels. These levels correlate positively with bone formation markers (type I procollagen peptide, alkaline phosphatase, and osteocalcin) and are crucial for bone synthesis. Consequently, patients with type 1 diabetes (T1D), characterized by low IGF-1 levels, often develop osteoporosis and exhibit increased fracture risk [5]. Bouillon [6] further links low serum IGF-1 levels to bone loss in T1DM patients. The development of diabetic Charcot neuroarthropathy, primarily driven by autonomic neuropathy under the ‘neurovascular theory,’ involves impaired veno-arterial shunting and enhanced arterial perfusion, leading to increased local blood flow [7]. This heightened flow, coupled with elevated calcium dissolution, intensifies osteoclastic activity—particularly through the receptor activator of nuclear factor kappa-B ligand (RANK-L) [8]—promoting bone demineralization, structural degradation, and subsequent fractures or foot ulcers [9]. Alternatively, the ‘neurotraumatic theory’ posits that peripheral neuropathy (distal sensorimotor polyneuropathy) results in sensory loss and painless, recurrent minor joint trauma [10]. In type 1 diabetics with low bone mass, this susceptibility to fractures initiates a pathological cascade culminating in joint destruction.
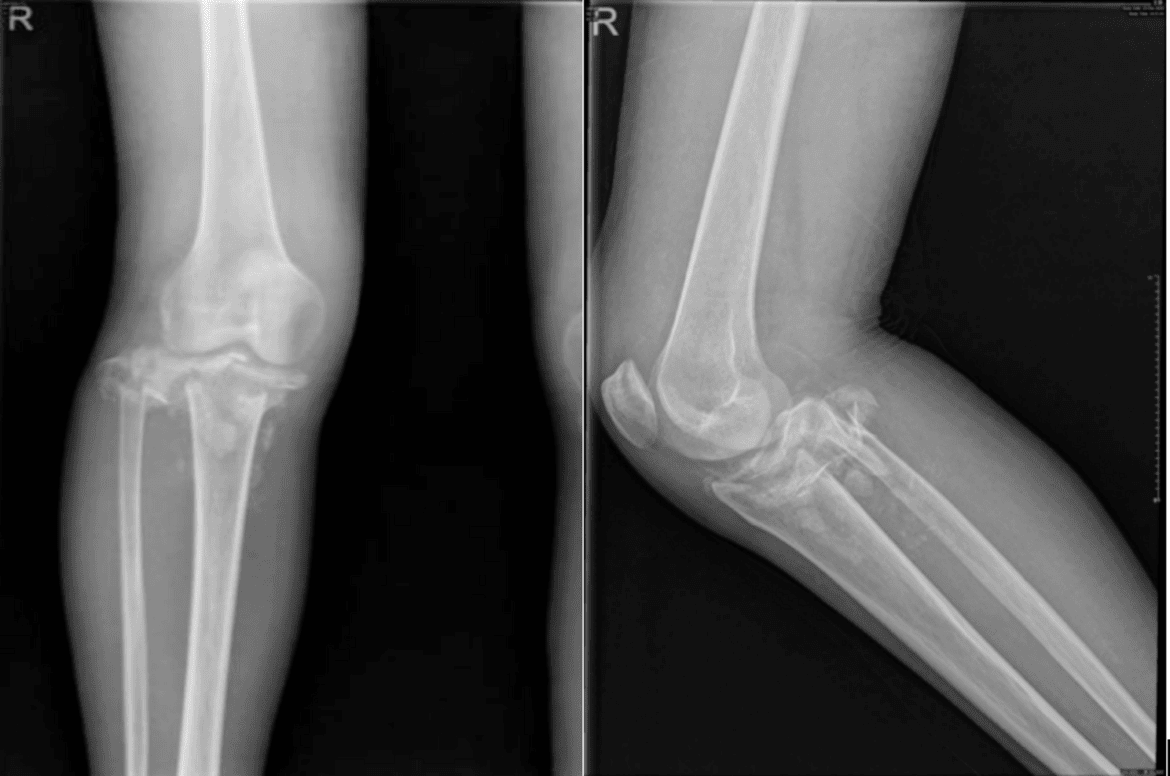
Literature indicates that type 1 diabetes mellitus (T1DM) typically manifests during adolescence [11]frequently presenting significant glycemic control challenges that precipitate complications including reduced bone density and neurovascular damage. Younger patients exhibit heightened susceptibility to neuroarthropathy due to greater physical activity demands. Recent years show increased recognition of diabetic Charcot neuroarthropathy (CN) affecting the knee joint. The condition’s nonspecific and often painless presentation frequently leads to early misdiagnosis, ultimately resulting in severe joint destruction and disability. Consequently, early detection and intervention for medial compartment imaging abnormalities in T1DM patients—particularly those with suboptimal glycemic control—are critical to prevent progressive joint deterioration.
In early-stage disease, non-surgical treatments such as plaster casting or knee bracing, combined with avoidance of weight-bearing, are used to prevent severe joint destruction and instability. Plaster casting typically lasts 12–18 weeks, with early intervention and adherence to partial weight-bearing significantly reducing healing time [12]. Patel [13] reported two cases of diabetic knee joint Charcot neuroarthropathy. In one case, a knee brace and gradual weight-bearing resulted in resolution of femoral condyle bone marrow edema within six months and complete disappearance of clinical and radiological symptoms by 13 months. The other case, treated with long leg plaster casting, showed no radiological healing and developed knee joint deformity with significant structural damage over four years, likely due to advanced disease stage (Table 2). In the second case we report, initial treatment with a knee brace led to significant medial tibial plateau collapse fracture and unstable joint movement on five-month follow-up X-ray, ultimately necessitating knee joint replacement surgery given the patient’s high activity level.
Pharmacotherapy targeting anti-resorption and bone reconstruction is gaining attention as an adjunct treatment for diabetic Charcot neuroarthropathy, particularly post-surgery to mitigate complications such as periprosthetic fractures and progressive bone loss. Bisphosphonates (BPs), followed by calcitonin, are frequently used for their anti-resorptive properties in conditions of increased bone turnover [14]. Despite the lack of definitive dosage regimens and long-term outcome data, BPs represent potential future options for both non-surgical and post-surgical adjunct therapy [15]. In a 12-month randomized controlled double-blind trial, Jude et al. ‘s [16] reported significant improvements with BP therapy, as evidenced by clinical and biochemical markers. Bem’s randomized controlled trial involving 32 acute diabetic Charcot neuroarthropathy patients demonstrated that intranasal calcitonin (200 IU daily) combined with oral calcium significantly reduced bone-specific alkaline phosphatase levels at three months compared to the control group. However, Pakarinen et al. ‘s [17] suggested that while BPs may reduce osteoclast activity and bone resorption, they could inadvertently prolong the disease course by delaying dead bone clearance. Teriparatide, a synthetic anabolic agent, is increasingly utilized for its osteoblast-stimulating effects, which enhance trabecular and cortical bone formation and improve bone microstructure [18]. Tamai et al. ‘s [19] reported successful treatment of Charcot arthropathy with teriparatide in a case of non-union following multiple failed ankle fusion surgeries. Rastogi et al.‘s [20] clinical trial further demonstrated teriparatide’s capacity to promote new bone formation, elevate P1NP bone formation markers, and increase bone density. However, its efficacy in knee joint Charcot neuroarthropathy requires validation in subsequent clinical trials. Denosumab, a human monoclonal antibody against RANK-L approved for osteoporosis and malignant bone resorption, represents a rational therapeutic approach for Charcot joint disease. Carvès et al. ‘s [21] observed significant symptomatic relief and delayed joint destruction in seven patients after 16 months of denosumab treatment.
Surgical intervention—including knee arthrodesis, replacement, or amputation—is the preferred treatment for late-stage knee joint Charcot neuroarthropathy. Total knee arthroplasty (TKA), once contraindicated due to high rates of serious postoperative complications (e.g., periprosthetic fractures, aseptic loosening, instability, and infection [22]), particularly in diabetic patients, is now reconsidered owing to advances in prosthetic materials. Recent studies suggest improved outcomes with knee replacement in such cases. The choice of prosthesis type and fixation method is critical: non-constrained and short-stem prostheses may fail prematurely in osteoarthropathy of neurological origin. Severely unstable knees, irrespective of neurological involvement, require rotating-hinge or constrained prostheses for inherent stability, though these increase stress at the bone-cement-implant interface, elevating aseptic loosening risk [23]. Kim et al. [24] reported satisfactory outcomes in >50% of 19 neurosyphilitic Charcot joint patients treated with bone cement and constrained condylar prostheses, despite a 44% complication rate. Advances in prosthetic materials may enable less restrictive designs, reducing early loosening. Troyer et al. [25] successfully treated a Charcot joint patient using minimally constrained TKA with porous tantalum cones for structural support, demonstrating newer approaches’ potential. Lambert et al. [26] documented a 4-year complication-free outcome in a Type 1 diabetic patient who underwent TKA after failed conservative treatment. Conversely, Goetti et al. [27] reported a controversial case of bilateral knee Charcot neuroarthropathy treated with rotating-hinge prostheses, where postoperative periprosthetic fractures necessitated 6 months of teriparatide therapy, highlighting evolving strategies.
In late-stage knee joint Charcot neuroarthropathy, significant bone loss and defects present surgical challenges. During knee replacement, constrained prostheses with distal fixation aid in reconstructing normal joint morphology and restoring bone mass. Treatment options for bone defects include cement augmentation, metal tantalum blocks [28, 29]and bone grafting [30]. Date et al. [31] successfully treated a Type 1 diabetic patient with allogeneic bone grafting following a partial proximal tibial medial fracture, demonstrating this approach’s efficacy. Kim et al. [24] emphasized postoperative activity restrictions to promote graft remodeling and prevent early dislocation. In a second reported case, allogeneic impaction bone grafting reinforced with titanium mesh and screws yielded favorable outcomes without significant resorption. Our surgical team routinely employs this technique for severe hip joint defects with consistent success, further validating its utility.