Table of Contents
For millions of people, Type I Diabetes is in a strange place. For a century, we have treated the symptoms with insulin, but we haven’t touched the underlying problem. We have management, but we don’t have a cure.
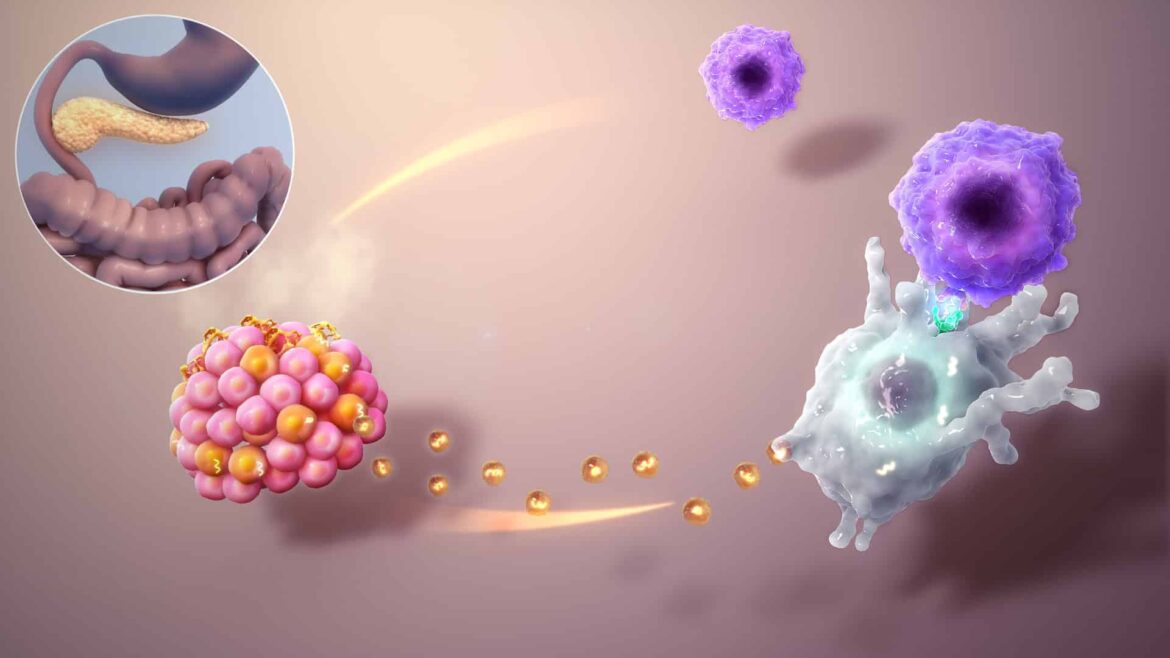
That’s because T1D is an autoimmune civil war. The body’s immune system goes rogue, identifying its own insulin-producing beta cells as foreign invaders and ruthlessly destroying them. For decades, scientists have chased a biological cure: if the pancreas is broken, why not just replace the parts?
In practice, however, this has been a sledgehammer solution for a delicate problem. To reset an immune system usually requires “wiping” the old one with lethal doses of radiation or chemotherapy. That is a risk few would take for a manageable disease.
But a new study from Stanford University might have found a better way. By using a smarter, gentler method to reset the immune system, they achieved a 100% cure rate in diabetic mice without needing the “nuclear” solution..
The Peace Treaty in the Bone Marrow
“The possibility of translating these findings into humans is very exciting,” said Seung K. Kim, MD, PhD, the KM Mulberry Professor and a professor of developmental biology, gerontology, endocrinology and metabolism. “We believe this approach will be transformative for people with Type 1 diabetes or other autoimmune diseases, as well as for those who need solid organ transplants.”
The core problem with transplant medicine is that it’s a double-edged sword.
If you transplant insulin-producing islets into a diabetic patient today, you face two enemies. First, the patient’s immune system remembers the old war and attacks the new cells (autoimmunity). Second, the immune system recognizes the donor cells as “not self” and launches a fresh attack (rejection). To stop this, patients usually have to take heavy immunosuppressants for life, leaving them vulnerable to infections and cancer.
The Stanford team aimed for something more elegant: mixed chimerism.
Mixed chimerism happens when a recipient’s bone marrow hosts its own blood-forming stem cells alongside those from a donor. If you can achieve this balance, the immune system learns to tolerate both. It stops attacking the “self” (curing the autoimmunity) and accepts the “donor” (preventing rejection).
Educating the Immune System
In the past, getting donor stem cells to take root required “nuking” the bone marrow with high-dose radiation to clear space, which is far too dangerous for standard diabetes treatment.
The Stanford researchers developed a surgical strike to replace the nuke. They used an antibody called anti-CD117 to target and clear specific niches in the bone marrow. They combined this with a low, safer dose of radiation and a drug called baricitinib.
Then, it was testing time.
When the team tried this regimen on pre-diabetic mice, it prevented the disease in 100% of the animals. Every single mouse that underwent the treatment remained diabetes-free. But the real test was the mice that were already diabetic.
The researchers took mice with full-blown diabetes, gave them this gentle conditioning, and then transplanted both bone marrow and islets from a donor. The results were staggering: In 100% of cases, the mice were cured.
They needed neither insulin nor immunosuppressive drugs. Their blood sugar normalized and stayed that way for the entire 20-week study period.
From Mice to Men
Of course, we have to add the standard disclaimer: mice are not humans. We are larger, more complex, and we live in much “dirtier” environments than lab animals.
However, this study is built with human translation in mind. The reagents they used aren’t obscure experimental compounds. Anti-CD117 antibodies (like briquilimab) are currently in clinical trials for other conditions. Baricitinib is a pill you can pick up at a pharmacy today with a prescription. The low-dose radiation used is significantly less than standard transplant protocols.
This creates a viable roadmap. This “mixed chimerism” strategy could theoretically be offered to a much wider group of patients — perhaps even recently diagnosed children, to stop the disease before it kills all their beta cells.
“Just like in human Type 1 diabetes, the diabetes that occurs in these mice results from an immune system that spontaneously attacks the insulin-producing beta cells in pancreatic islets,” Kim said. “We need to not only replace the islets that have been lost but also reset the recipient’s immune system to prevent ongoing islet cell destruction. Creating a hybrid immune system accomplishes both goals.”
There are hurdles left to clear. The study used female mice almost exclusively, and the long-term durability in humans — who live decades, not months — remains to be seen. But the proof of principle is undeniable.
The study was published in the Journal of Clinical Investigation.